BAPETEN Conducts Operational License Verification for Radioisotope and Radiopharmaceutical Production Facility of PT Kardia Farma Solusindo
Kembali 30 April 2026 | Berita BAPETEN | 24 lihatBAPETEN, through the Directorate of Licensing of Radiation Facilities and Radioactive Materials (DPFRZR), conducted an operational license verification for the radioisotope and radiopharmaceutical production facility of PT Kardia Farma Solusindo in East Jakarta from 27 to 30 April 2026. The activity was carried out to ensure that the facility meets radiation safety and radioactive source security requirements in order to protect workers, patients, the public, and the environment.
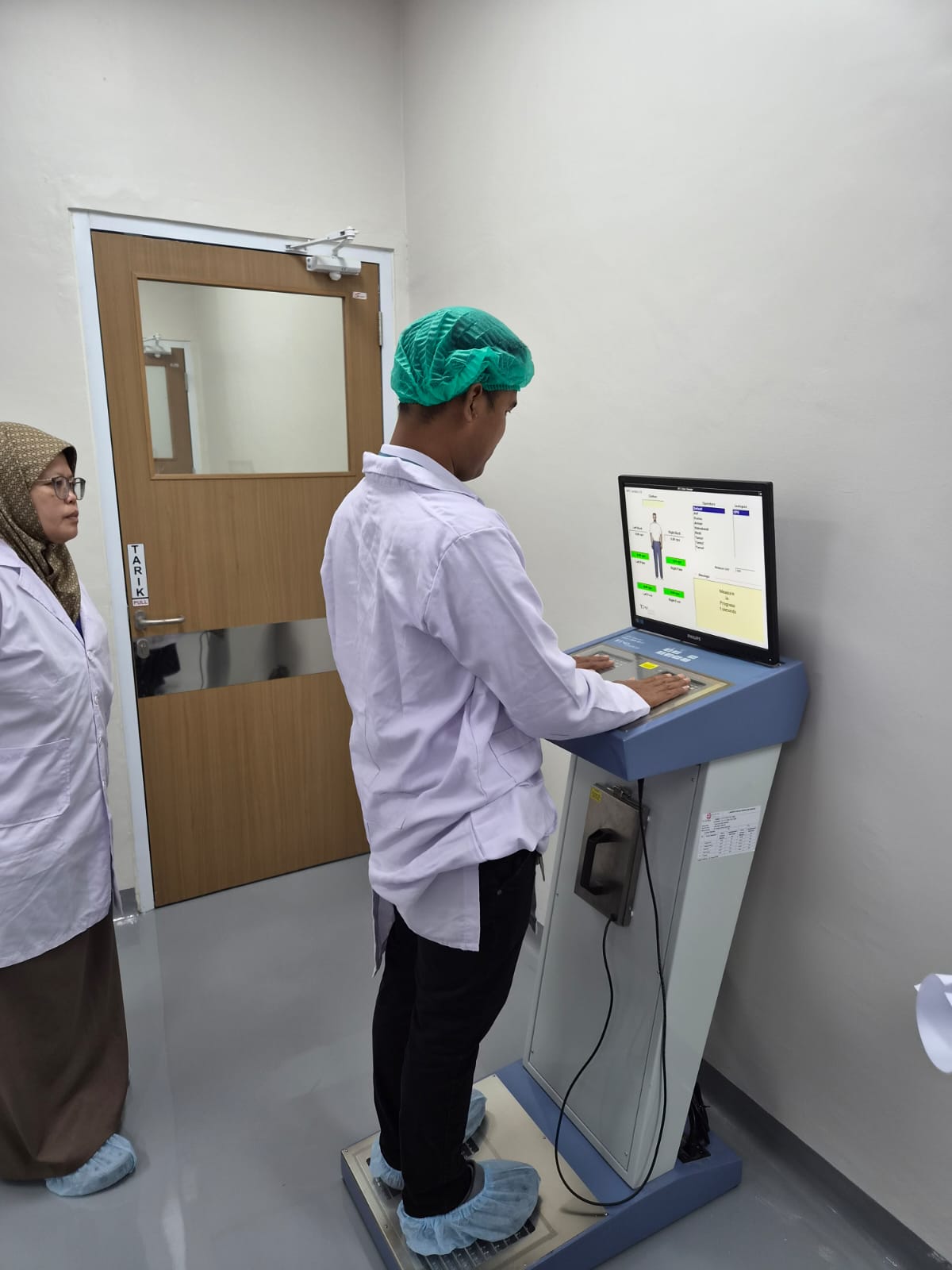
The verification covered the operation of a radioisotope and radiopharmaceutical production facility utilizing the IBA Cyclone® KIUBE 300 cyclotron for the production of F-18 Fluorodeoxyglucose (FDG). The inspection team was led by Made Pramayuni, Senior Radiation Inspector of the Health Facility Licensing Functional Group at DPFRZR BAPETEN, together with members of the verification team.
During the onsite verification, the team conducted a comprehensive review of documents and technical aspects, including the conformity of the constructed facility with the approved design, adequacy of radiation shielding in the cyclotron bunker, and readiness of the facility’s main and supporting equipment. The inspection also covered target material receiving and storage areas, cyclotron facilities, radiopharmaceutical production facilities, commissioning tests, bombardment tests for radioisotope production, and synthesis processes conducted in the hot cell. In addition, the team verified quality control procedures for radioisotope and radiopharmaceutical products, radiation safety systems, cyclotron operating systems, radiation exposure measurements within the facility, waste management systems for solid, liquid, and gaseous waste, radiation protection equipment, supporting instruments, and personnel readiness.
Based on the verification results, PT Kardia Farma Solusindo committed to completing all findings and recommendations provided by the BAPETEN team within the specified timeframe. The company also expressed its appreciation for the verification process and viewed the findings as valuable technical input to further strengthen compliance with applicable radiation safety regulations and standards.
PT Kardia Farma Solusindo further expressed hope that the operation of its radiopharmaceutical production facility, particularly for the production of Fluorodeoxyglucose (FDG), would contribute to expanding access to healthcare services for cancer patients in Indonesia by supporting more accurate, accessible, and affordable diagnostic services through high-quality radiopharmaceutical products. [DPFRZR/Dwiangesti/BHKK/YL/HRUF]

























Komentar (0)